New drugs to treat COVID-19 approved by EMA
The European Medicines Agency (EMA) has approved two monoclonal antibodies, Regkirona (regdanvimab) and Ronapreve (casirivimab / imdevimab), as biological drugs in the fight against COVID-19.
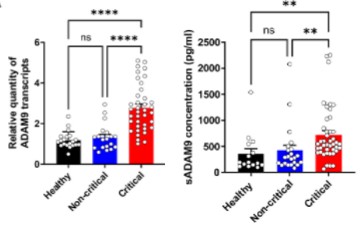
Identification of driver genes for critical forms of COVID-19 in a deeply phenotyped young patient cohort
Research using multi-omics analysis (whole genome sequencing, RNA sequencing, proteomics, cytokine profiling and high-throughput immunophenotyping) combined with artificial intelligence has shown that increased level of ADAM9
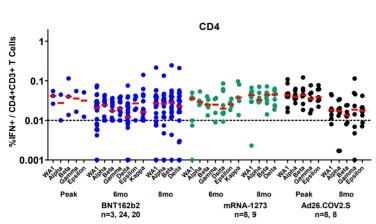
Differential Kinetics of Immune Responses Elicited by Covid-19 Vaccines
A study published in NEJM shows how the cellular and humoral response changes within 8 months of receiving either the two-dose BNT162b2 or mRNA-1273 vaccine or the single dose Ad26.COV2.S vaccine
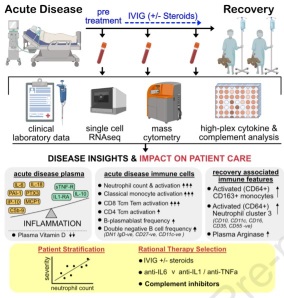
The immune landscape of SARS-CoV-2-associated Multisystem Inflammatory Syndrome in Children (MIS-C) from acute disease to recovery
Multisystem Inflammatory Syndrome in Children (MIS-C / PIMS) is a life-threatening disease that occurs several weeks after SARS-CoV-2 infection. High-throughput immune profiling showed that patients with acute MIS-C had significantly elevated levels of neutrophils
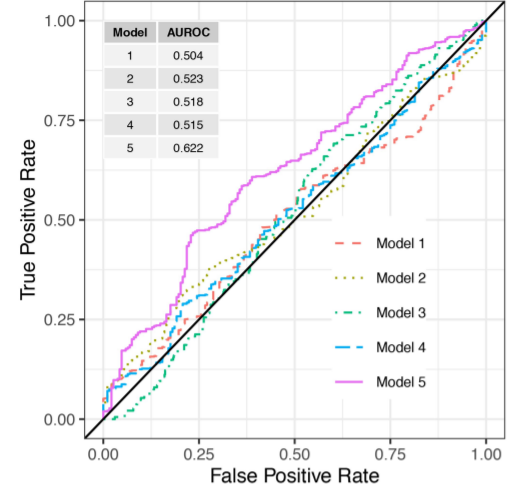
Efficient and targeted COVID-19 border testing via reinforcement learning
Research published in Nature presents a machine learning algorithm called 'Eva' to limit the influx of tourists infected with SARS-CoV-2 into Greece.
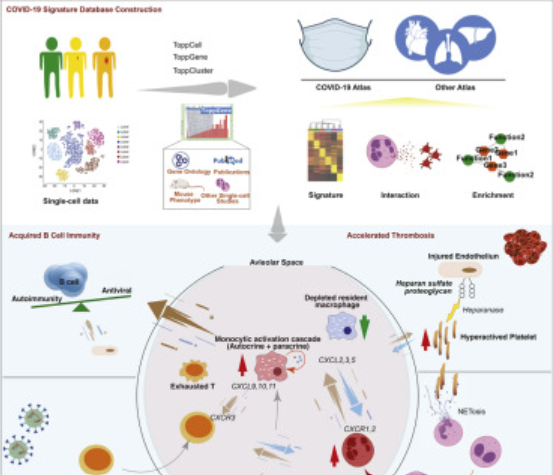
An Interactive Single Cell Web Portal Identifies Gene and Cell Networks in COVID-19 Host Responses
Scientists from US have created a new web portal (ToppCell: COVID-19 Atlas), thanks to which it is possible to compare the results of research from many research centers using scRNA-seq