COVID-19 vaccines in Poland
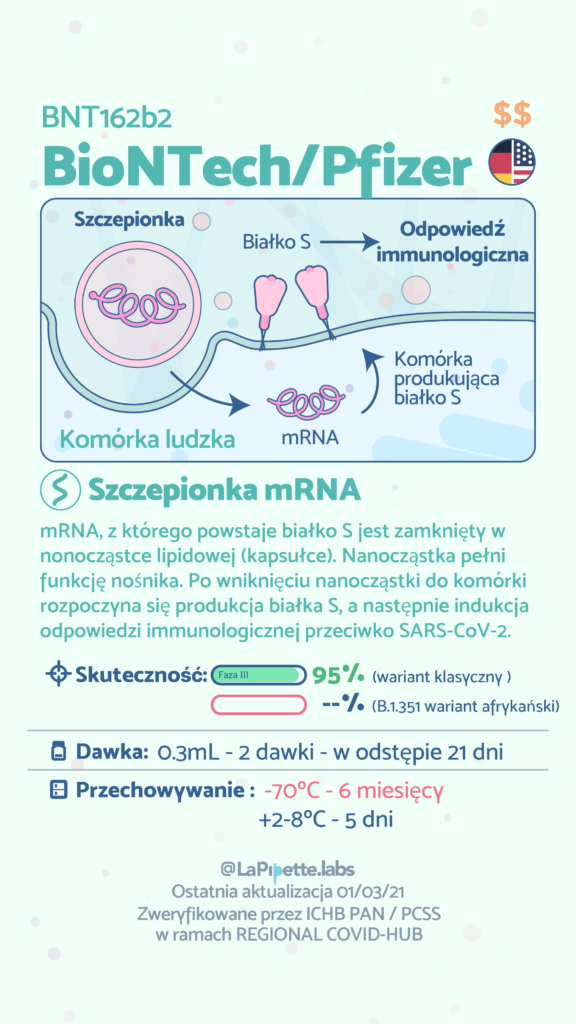
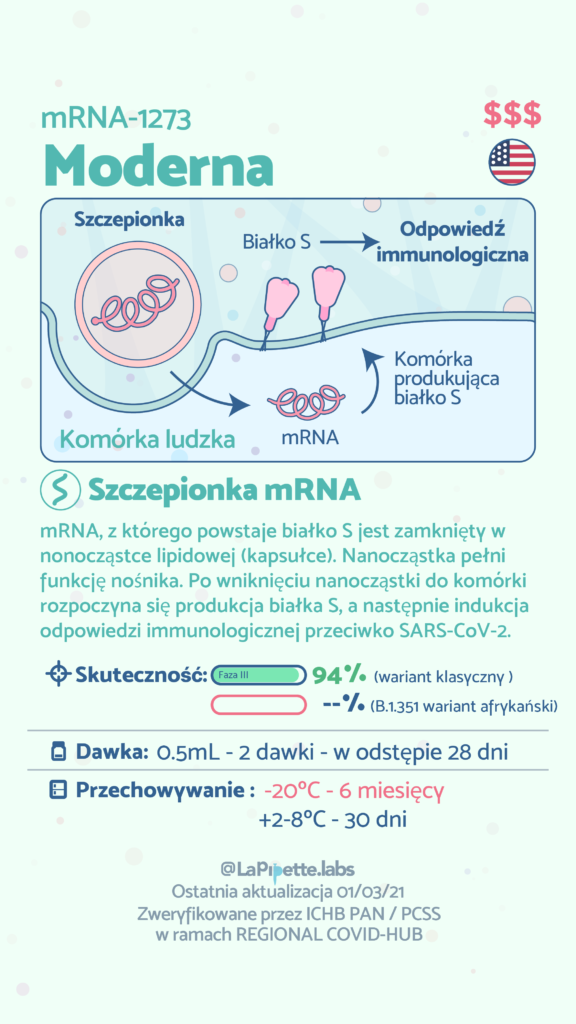
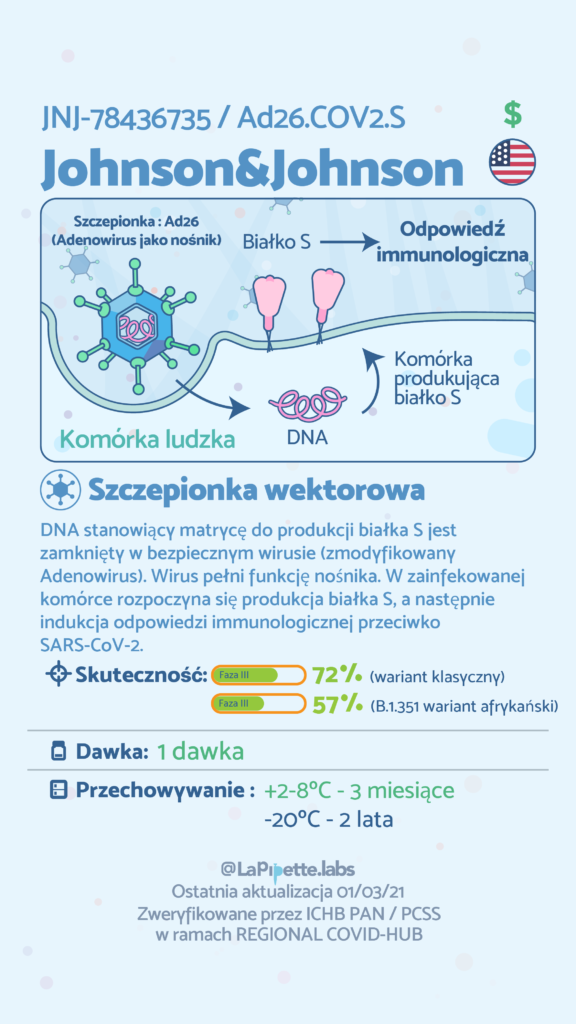
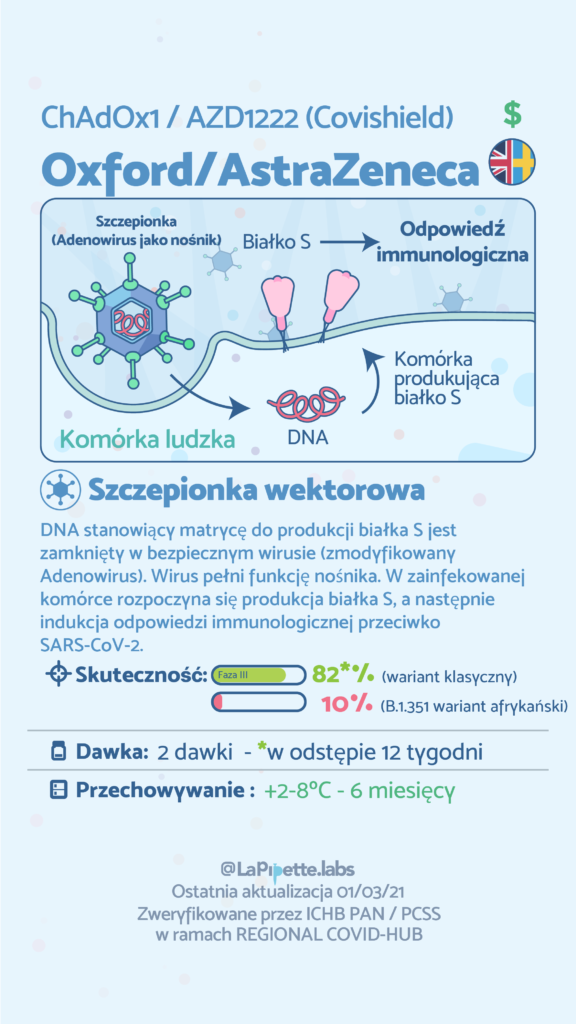
In Poland, COVID-19 vaccination started on December 27, 2020 as part of the National Vaccination Programme. So far, the programme uses four different products approved by the European Medicines Agency(EMA) containing mRNA or viral vectors. The COVID-19 vaccines are produced by pharmaceutical companies including BioNTech / Pfizer, Moderna, AstraZeneca and Janssen Pharmaceuticals (owned by Johnson & Johnson). The fifth product developed by Novavax is using recombinant SARS-CoV-2 proteins and was recently approved by EMA. It is expected that the production of the active substance of Novavax (ie viral proteins) will take place in Poland. At the same time, research has begun on the effectiveness of subsequent vaccines against new variants of SARS-CoV-2. As a result, in the future, the production of vaccines against the new variants will be possible in a short time and on a large scale.
Date of EMA approval of COVID-19 vaccines